We did this lab to help us learn how molecules are structured and how bonds occur. We were given gumdrops, raisins and toothpicks to make large scale molecules of several compounds to help understand how they are grouped and how electrons are shared.
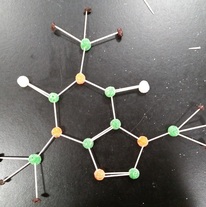
| Phosphorus trihydride. (PH3) 40 valence electrons Aluminum hydride.( ALH3 ) 6 valence electrons. formaldehyde ( CH2O ) 12 valence electrons. carbon disulfide (CS2) 16 valence electrons. nitrogen trifluoride (NF3) 26 valence electrons caffeine (C8H10N4O2) Nicotine C10H14N2 | This is phosphorus pentafluoride. (PF5) 40 valence electrons. sulfur dioxide (SO2) 18 valence electrons. sulfur trioxide (SO3) 24 valence electrons. Oxide (O3) 18 valence electrons. dihydrogen sulfide (H2S) 8 valence electrons Aspirin (C9H8O4) |












 RSS Feed
RSS Feed
